P07: Epigenomic mapping of myogenic regulation in human muscle development
Open postions: 1 Postdoctoral researcher and 1 PhD student
Principal investigators: Prof. Dr. Uwe Ohler and Assistant Prof. Dr. Ferah Yildirim
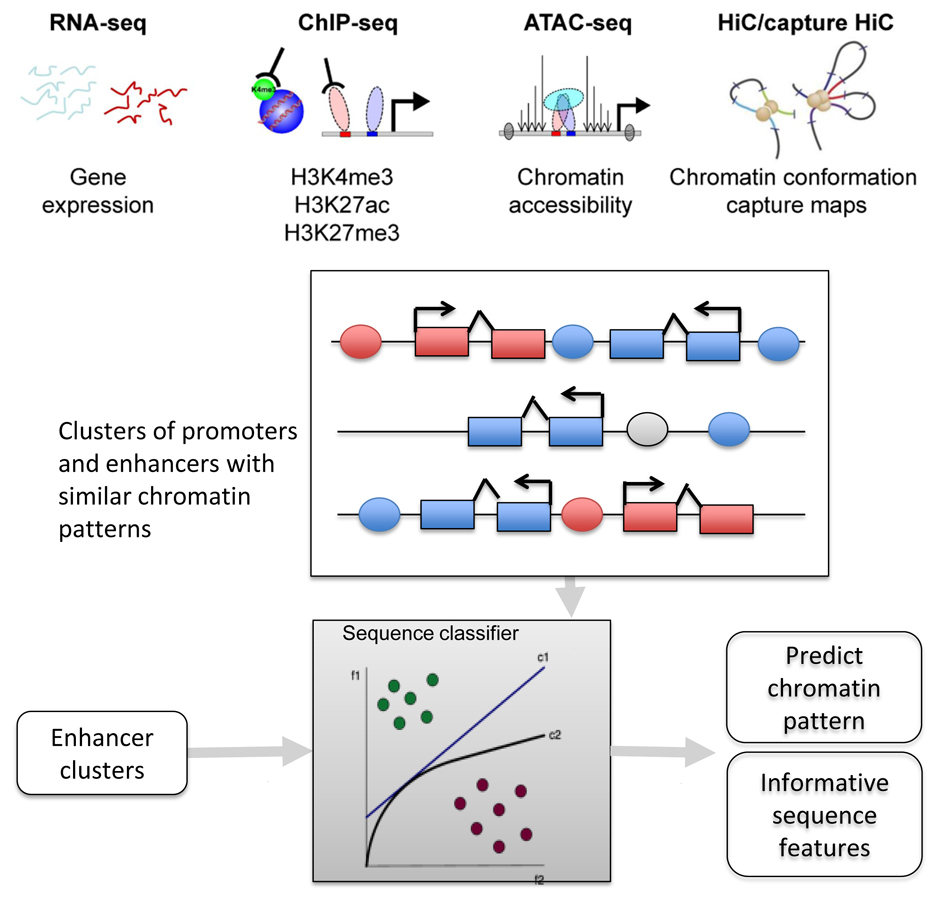 Congenital myopathies are a group of genetic muscle disorders clinically characterized by muscle hypotonia and weakness and a static or slowly progressive clinical course. Widespread use of whole exome sequencing (WES) has led to the identification of the underlying genetic cause of many different congenital myopathies over the course of the last decade, but a large subset of these cases have eluded all attempts at finding a molecular diagnosis. The Charité has amassed a cohort of congenital myopathy patients where WES had failed to identify a genetic cause in protein coding sequences. We hypothesize that the disease-causing variants in this cohort are located in the non-coding or regulatory sequences of the genome. These regions, which harbor enhancers and transcription factor binding sites (TFBSs), control muscle-specific gene expression programs, whose dysfunction is at the root of their pathogenesis. Though whole genome sequencing (WGS) is now technically able to uncover structural and single nucleotide variants and indels in the non-coding genome, we are in the need of more information about the epigenetic regulation that governs muscle development in order to interpret these variants. Our collaborative research program will start by conducting a series of comprehensive epigenomic and transcriptional analyses of consecutive developmental cell stages along the muscle cell differentiation trajectory in cellular model systems derived from established iPSC lines. This information will be used to train computational models for different enhancer subgroups that will be used to identify relevant DNA sequence variation within myogenic regulatory regions predicted to alter the transcriptional programs in patients with congenital myopathy. We will continue to evaluate the pathogenicity of such non-coding variants in relevant cell and animal models. The approaches developed and tested here are integral to the success of the overall RU goals, specifically for the translation of the basic molecular biology into diagnoses and therapies for patients with rare muscle disorders.
Congenital myopathies are a group of genetic muscle disorders clinically characterized by muscle hypotonia and weakness and a static or slowly progressive clinical course. Widespread use of whole exome sequencing (WES) has led to the identification of the underlying genetic cause of many different congenital myopathies over the course of the last decade, but a large subset of these cases have eluded all attempts at finding a molecular diagnosis. The Charité has amassed a cohort of congenital myopathy patients where WES had failed to identify a genetic cause in protein coding sequences. We hypothesize that the disease-causing variants in this cohort are located in the non-coding or regulatory sequences of the genome. These regions, which harbor enhancers and transcription factor binding sites (TFBSs), control muscle-specific gene expression programs, whose dysfunction is at the root of their pathogenesis. Though whole genome sequencing (WGS) is now technically able to uncover structural and single nucleotide variants and indels in the non-coding genome, we are in the need of more information about the epigenetic regulation that governs muscle development in order to interpret these variants. Our collaborative research program will start by conducting a series of comprehensive epigenomic and transcriptional analyses of consecutive developmental cell stages along the muscle cell differentiation trajectory in cellular model systems derived from established iPSC lines. This information will be used to train computational models for different enhancer subgroups that will be used to identify relevant DNA sequence variation within myogenic regulatory regions predicted to alter the transcriptional programs in patients with congenital myopathy. We will continue to evaluate the pathogenicity of such non-coding variants in relevant cell and animal models. The approaches developed and tested here are integral to the success of the overall RU goals, specifically for the translation of the basic molecular biology into diagnoses and therapies for patients with rare muscle disorders.