P01: The role of dynamic gene expression in myogenic stem cells
Open postions: 1 Postdoctoral researcher
Principal investigator: Prof. Dr. Carmen Birchmeier
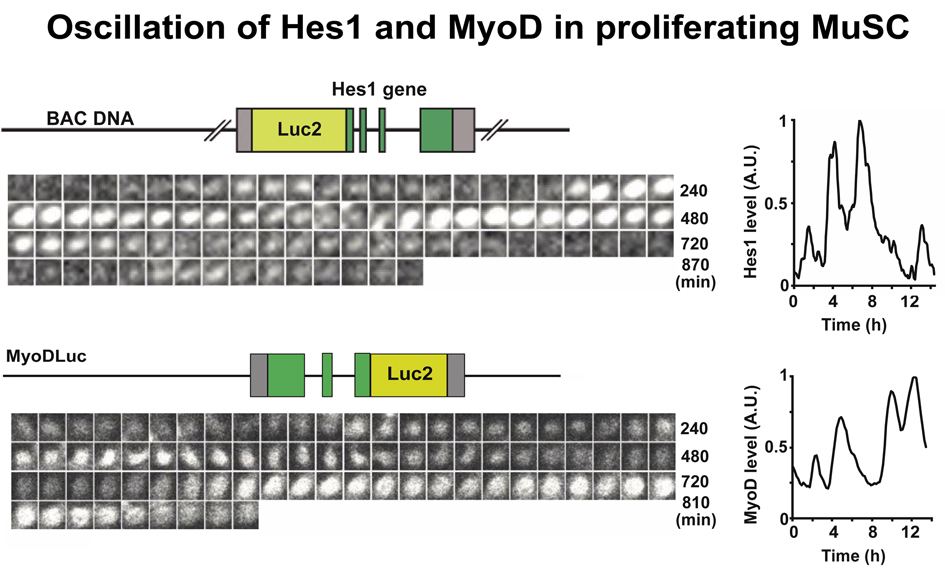 The developing muscle forms and maintains a pool of myogenic stem cells. These resident stem cells are the source of myoblasts for muscle growth during development and generate satellite cells in the perinatal period. Deficits in the maintenance, proliferation, and differentiation of muscle stem cells play an important role in inherited muscle disorders. Emerging evidence indicates that the expression dynamics of regulatory molecules and their fluctuation control the maintenance and differentiation of stem cells, thereby controlling stem cell fate. Our unpublished work shows that the transcription factors Hes1 and MyoD display oscillatory expression in proliferating muscle stem cells. The oscillatory period is around 3 hours, much shorter than the oscillations of the cell cycle or the circadian rhythm. Moreover, oscillations are asynchronous and neighboring cells oscillate out of phase. Oscillatory expression of Hes1 drives the oscillatory expression of MyoD. Ablation of the Hes1 oscillator dampens MyoD oscillations, causes sustained MyoD expression, and increases a cell’s tendency to differentiate. This leads to marked deficits in the maintenance of muscle stem cells, resulting in impaired muscle growth and repair. The overarching goal of this proposal is to define the network of oscillatory genes in muscle stem cells, to assess the consequences of MyoD/Hes1 oscillations on a mechanistic level, and to apply this information to the identification of disease-causing variants in these networks in patients with disorders of muscle development. We will use optogenetic tools to synchronize transcription factor (TF) oscillations and define gene expression during different phases of the oscillatory cycle. This will reveal the network of genes that are expressed in an oscillatory manner. Optogenetic tools will be used to assess whether sustained or oscillatory expression of MyoD/Hes1 directly influences gene expression and the chromatin state. Insight into such oscillatory cycles during stem cell maintenance and differentiation, into their epigenetic regulation, and their computational modeling will help us identify genes mutated in congenital myopathies and other disorders of human muscle development.
The developing muscle forms and maintains a pool of myogenic stem cells. These resident stem cells are the source of myoblasts for muscle growth during development and generate satellite cells in the perinatal period. Deficits in the maintenance, proliferation, and differentiation of muscle stem cells play an important role in inherited muscle disorders. Emerging evidence indicates that the expression dynamics of regulatory molecules and their fluctuation control the maintenance and differentiation of stem cells, thereby controlling stem cell fate. Our unpublished work shows that the transcription factors Hes1 and MyoD display oscillatory expression in proliferating muscle stem cells. The oscillatory period is around 3 hours, much shorter than the oscillations of the cell cycle or the circadian rhythm. Moreover, oscillations are asynchronous and neighboring cells oscillate out of phase. Oscillatory expression of Hes1 drives the oscillatory expression of MyoD. Ablation of the Hes1 oscillator dampens MyoD oscillations, causes sustained MyoD expression, and increases a cell’s tendency to differentiate. This leads to marked deficits in the maintenance of muscle stem cells, resulting in impaired muscle growth and repair. The overarching goal of this proposal is to define the network of oscillatory genes in muscle stem cells, to assess the consequences of MyoD/Hes1 oscillations on a mechanistic level, and to apply this information to the identification of disease-causing variants in these networks in patients with disorders of muscle development. We will use optogenetic tools to synchronize transcription factor (TF) oscillations and define gene expression during different phases of the oscillatory cycle. This will reveal the network of genes that are expressed in an oscillatory manner. Optogenetic tools will be used to assess whether sustained or oscillatory expression of MyoD/Hes1 directly influences gene expression and the chromatin state. Insight into such oscillatory cycles during stem cell maintenance and differentiation, into their epigenetic regulation, and their computational modeling will help us identify genes mutated in congenital myopathies and other disorders of human muscle development.